The UF/IFAS Honey Bee Research and Extension Laboratory conducted research projects that led to two 2020 publications on Varroa destructor. Below are summaries of the research as well as links to the full publications.
Rearing Varroa destructor in the laboratory
Purpose: The purpose of this research project was to evaluate the efficacy of different methods of rearing Varroa in vitro. As Varroa continue to impact honey bee populations negatively worldwide, the development of new and innovative mite control methods should remain a priority among honey bee researchers.
Key Findings:
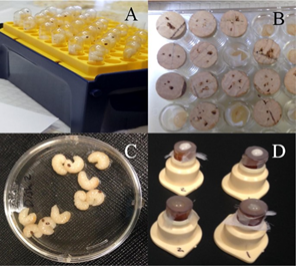
- Four methods were tested for rearing Varroa destructor in vitro (being kept alive in a laboratory rather than in a honey bee hive). Varroa survived best in gelatin capsules that contained a honey bee pupa, on which the mites could feed and reproduce.
- The investigative team concluded that the best conditions to keep Varroa alive in the laboratory were at 75% humidity and in 7-mm diameter gelatin capsules.
Methods of rearing Varroa in vitro. A. Honey bee prepupae and Varroa females placed inside 7-mm (outside diameter) gelatin capsules. The capsules were placed vertically in an empty micro-pipette tip box. B. Varroa were placed in wells containing in vitro reared honey bee prepupae. The wells were closed with cork plugs. C. Ten Varroa and 10 worker honey bee prepupae were placed in a sterile Falcon 50 × 9 mm Petri dish. In this method, the mites were able to feed on any of the developing honey bees. d Rearing chambers created based on the Bruce et al. (1988) design to test the feasibility of feeding honey bee hemolymph or fat tissue to Varroa. The chambers in the top row contain fat tissue from dissected honey bee prepupae placed on the Parafilm membrane. Those on the bottom row contain honey bee prepupae hemolymph placed on the membrane. Images taken from Jack et al. 2020.
Significance: The findings from this research eliminates three methods that are unlikely to be successful for future in vitro rearing attempts and identifies a single method as the one most likely to be successful. Rearing Varroa in vitro has the potential to increase and improve Varroa control research. These findings also provide valuable information on maintaining Varroa in the laboratory.
For more details, read the publication here: https://link.springer.com/article/10.1007/s10493-020-00488-0
Jack, C.J., Dai, P., van Santen, E., Ellis, J.D. 2020. Comparing four methods of rearing Varroa destructor in vitro. Experimental and Applied Acarology, 80(4): 463-476. https://doi.org/10.1007/s10493-020-00488-0.
Oxalic Acid (OA) and Brood Interruption in Controlling Varroa
Purpose: The purpose of this research project was to evaluate the efficacy of OA vaporization treatment as a Varroa control in honey bee hives.
Key Findings:
- OA, an organic compound found in plants, was vaporized and administered to hives per label directions. Brood interruption was administered to some colonies by caging a queen for 24 days, removing all brood from the nests. The researchers found that colonies treated with OA vaporization at the current legal rate of 1 g OA per brood chamber experienced higher mortality rates and lower overall health than colonies treated with Apivar®️, the current industry standard. Additionally, brood interruption is not a recommended practice in the late summer in Florida, as many colonies did not recover fully from the dramatic loss of brood.
- In conclusion, neither OA nor brood disruption provided adequate Varroa control.
Significance: These findings provide valuable information related to Varroa control using OA vaporization and brood interruption.
For more details, read the publication here: https://academic.oup.com/jee/article-abstract/113/2/582/5697464?redirectedFrom=fulltext
Jack, C.J., van Santen, E., Ellis, J.D. 2020. Evaluating the efficacy of oxalic acid vaporization and brood interruption in controlling the honey bee pest Varroa destructor (Acari: Varroidae). Journal of Economic Entomology, 113(2): 582-588. https://doi.org/10.1093/jee/toz358.
This blog was written by UF HBREL graduate student, Brynn Johnson
 0
0